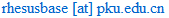Rhesus Macaques DNA Swap may Prevents Mitochondrial Diseases
2009-08-26

Twin rhesus macaques "Mito" and "Tracker" were born from transplanted DNA.
Credit: Oregon Health & Science University
As reported in the online edition of Nature on August 26, 2009, Dr.Shoukhrat Mitalipov and his colleagues at the Oregon Health & Science University have developed a new technique to safety remove nuclear DNA from monkey egg cells without bringing along any mitochondrial DNA. This method provides new options for preventing certain inherited disorders that pass from mother to child in mitochondria, and may be used to prevent certain genetic disease.
For details, refer to the publication:
Mitochondrial gene replacement in primate offspring and embryonic stem cells. Nature (2009) doi:10.1038/nature08368.Masahito Tachibana, Michelle Sparman, Hathaitip Sritanaudomchai, Hong Ma, Lisa Clepper, Joy Woodward,Ying Li, Cathy Ramsey, Olena Kolotushkina& Shoukhrat Mitalipov
Abstract:
Mitochondria are found in all eukaryotic cells and contain their own genome (mitochondrial DNA or mtDNA). Unlike the nuclear genome, which is derived from both the egg and sperm at fertilization, the mtDNA in the embryo is derived almost exclusively from the egg; that is, it is of maternal origin. Mutations in mtDNA contribute to a diverse range of currently incurable human diseases and disorders. To establish preclinical models for new therapeutic approaches, we demonstrate here that the mitochondrial genome can be efficiently replaced in mature non-human primate oocytes (Macaca mulatta) by spindle-chromosomal complex transfer from one egg to an enucleated, mitochondrial-replete egg. The reconstructed oocytes with the mitochondrial replacement were capable of supporting normal fertilization, embryo development and produced healthy offspring. Genetic analysis confirmed that nuclear DNA in the three infants born so far originated from the spindle donors whereas mtDNA came from the cytoplast donors. No contribution of spindle donor mtDNA was detected in offspring. Spindle replacement is shown here as an efficient protocol replacing the full complement of mitochondria in newly generated embryonic stem cell lines. This approach may offer a reproductive option to prevent mtDNA disease transmission in affected families.